Wounds may become stalled at the inflammation phase, exhibiting low-grade inflammation for a long period of time (sometimes indefinitely) due to numerous factors, both intrinsic and extrinsic (Table 1). Because of this, it is critically important to carry out a thorough holistic assessment of the wound, patient, wound management options and other treatments the patient may be undergoing. Understanding these factors is necessary for not only to initiate appropriate wound management, but also to ensure ancillary treatments are initiated to manage the underlying conditions.
Table 1. Factors that can delay or impair wound healing[1]
| Patient-related | Malnutrition (protein, carbohydrates, fatty acids and trace elements are essential for wound-healing) |
| Reduced peripheral blood supply (due to cardiovascular disorders, ischaemia) | |
| Obesity (decreases tissue perfusion) | |
| Underlying systemic condition (e.g. diabetes, autoimmune disorders) | |
| Impaired mobility/immobility | |
| Psychological stress, lack of sleep | |
| Patient non-compliance | |
| Other treatments | Medication (e.g. non-steroidal anti-inflammatory drugs, corticosteroids) |
| Chemotherapy | |
| Radiotherapy | |
| Wound-related | Microbial colonisation/infection |
| Maceration (e.g. due to high exudate volumes or contact with other moisture) | |
| Unrelieved pressure | |
| Service delivery | Inappropriate wound management |
| Reduced wound temperature (e.g. due to prolonged dressing changes or use of cold cleansing products) |
Haemostasis: Setting the stage for wound-healing
The presence of these factors can impair the functioning of microbiological processes that are needed to carry out wound-healing after haemostasis is achieved. Patients who have congenital or other impairment of platelet and fibrin production will take longer to achieve haemostasis, the first stage of wound healing, which begins as soon as the wound occurs.
Other, crucial compounds are released during this phase — including pro-inflammatory cytokines and growth factors, such as platelet-derived growth factor (PDGF), which stimulate epithelial cells, and recruit fibroblasts, neutrophils and monocytes — in preparation for the inflammatory phase of wound-healing[2].
The upside of inflammation
Excess, uncontrolled, long-term inflammation is considered one of the ‘classic’ signs of wound infection. And in these cases, it is problematic to wound-healing. Furthermore, when healing stalls, it is generally in the inflammation phase — which has led to inflammation being regarded as a ‘bad wolf’ in wound management. However, inflammation is a natural part of the healing process denoting activity at the site of repair — all those microbiological components are gathering at the wound to carry out their functions.
Neutrophils — a type of white blood cell responsible for ‘digesting’ and clearing cell debris and microorganisms, providing a first line of defence against infection[3–5]. In patients with compromised immune systems, these cells might not be adequately produced, and in those with compromised vascularity, insufficient numbers of neutrophils may not reach the wound site. Neutrophils have a further effect in that, as they die, they release enzymes such as elastase, which further digest debris and protect against microbial colonisation[3–5]. The underlying factors that impair neutrophil production and delivery should be addressed.
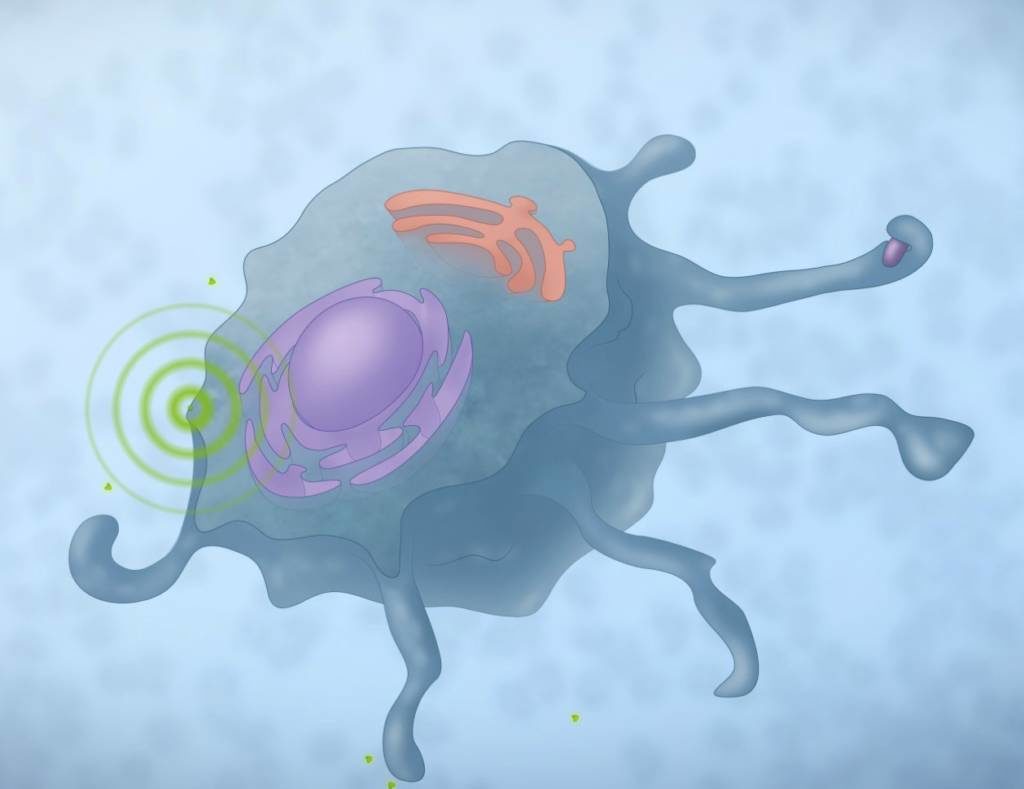
Macrophages — another type of white blood cell responsible for engulfing, digesting and clearing cell debris and microbial organisms, and considered the second line of defence against infection[3–5]. Macrophages are also considered the chief conductor in orchestrating the wound healing process itself, releasing factors necessary for pushing the process into the next stage of healing. Macrophages secrete matrix metalloproteases (MMPs), which are critical in autolytic debridement of necrotic tissue, as well as cytokines and growth factors — notably, fibroblast growth factor, epidermal growth factor, transforming growth factor-beta and interleukin-1 — that encourage granulation and epithelialisation[6].Using an advanced wound management product that activates macrophages can bolster treatment of underlying factors.
T-lymphocytes — these white blood cells are the third line, migrating to the wound after neutrophils and macrophages, which pave the way for their arrival[3–5]. Studies have found that delay of T-lymphocytes or inadequate concentrations correlate with stalled wound-healing[2]. Although studies have not yet pinpointed the precise causative function of T-lymphocytes in wound-healing, research suggests that some subsets of T-lymphocytes encourage certain cells critical for wound-healing, while other subsets suppress harmful cells[2,7,8].
Connecting the dots
Many of the factors that can delay wound healing — such as diabetes, malnutrition, impaired mobility, obesity and cardiovascular disorders — inhibit production, release and delivery of these cells. Macrophages, in particular, are imperative for the further production of cytokines and growth factors that help create healthy new tissue in the wound bed. Furthermore, if these white blood cells are not present or not performing their job in the wound bed, this can give rise to other factors that stall healing — such as microbial colonisation, excess wound exudate, and elevated levels of inflammatory cytokines, free radicals and proteases — creating a vicious-circle inflammation scenario[9].
Essentially, when a wound becomes stuck in a state of low-grade inflammation, it is due to microbiological impairments — the senescence or absence of cells needed for healing. These cells will usually respond to stimulation or activation with, for example, soluble beta-glucan, which can help attract the necessary cells to the wound bed and aid their effectiveness. Therefore, appropriate wound management — such as with Woulgan Bioactive Beta-Glucan Gel — is required. The active ingredient in Woulgan, soluble beta-glucan, helps activate macrophages which, in turn, restart normal production of desirable growth factors and attraction of cell-signalling molecules, to promote angiogenesis, cell proliferation and wound contraction.
References
- The Royal Children’s Hospital Melbourne. Clinical guidelines (nursing): Wound Care. Accessed March 2016 at: http://www.rch.org.au/rchcpg/hospital_clinical_guideline_index/Wound_care/#factors_delaying_healing
- Stacey M. Why don’t wounds heal? Wounds Int 2016;7(1):16–21
- Gosain A, DiPietro LA. Aging and wound healing. World J Surg 2004;28(3):321–6
- Broughton G 2nd, Janis JE, Attinger CE. The basic science of wound healing (retraction of Witte M, Barbul A. In: Surg Clin North Am 1997;77[3]:509–28). Plast Reconstr Surg 2006;117(7 Suppl):12S–34S
- Campos AC, Groth AK, Branco AB. Assessment and nutritional aspects of wound healing. Curr Opin Clin Nutr Metab Care 2008;11(3):281–8
- Field FK, Kerstein MD. Overview of wound healing in a moist environment. Am J Surg 1994;167(Suppl 1A):2S–6S
- Swift ME, Burns AL, Gray KL, DiPietro LA. Age-related alterations in the inflammatory response to dermal injury. J Invest Dermatol 2001;117(5):1027–35
- Park JE, Barbul A. Understanding the role of immune regulation in wound healing. Am J Surg 2004;187(5A):11S–16S
- Cullen B, Ivins N. PROMOGRAN & PROMOGRAN PRISMA Made Easy. Wounds Int 2010;1(3):S1–S6